Abstract
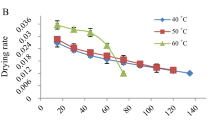
Costus pictus, recognized as the “insulin plant,” is a renowned medicinal plant with a diverse phytochemical composition, which confers a comprehensive spectrum of biological attributes. This is the first study on modeling the drying kinetics of Costus pictus rhizomes and analyzing the predictive capability of mathematical modeling and artificial neural network (ANN). C. pictus rhizomes were dried in a laboratory tray dryer at different temperatures (40 °C, 50 °C, and 60 °C) to produce a functional dried powder. Henderson and Pabis’s model well-described the drying kinetics. ANN with a feed-forward backpropagation algorithm was applied to understand the drying characteristics. Moisture ratio and moisture content were optimally trained with the Levenberg-Marquardt algorithm and hyperbolic tangent sigmoid transfer function for hidden layer. Comparative analysis validated that ANN had an unrivaled prediction potential. Color studies substantiated the overall acceptability of a dried product. Rhizomes dried at 60 °C had the best antioxidant, polyphenol, and flavonoid properties. Investigation into the influence of different solvents (water, ethanol, methanol, and ethyl acetate) on these attributes revealed that methanol, followed by water, showed most significant effects. Thus, 60 °C–dried rhizomes revealed shorter drying time with accelerated drying rates, and moisture diffusivity and activation energy were observed as 2.3520 × 10−7 m2/s and 41.088 kJ/mol, respectively. It also exhibited conserved qualitative characteristics and had high phytochemical constituents with remarkable antioxidant potential. The results provided a thorough grasp of the diverse traits of C. pictus rhizome powders, suggesting their utilization in herbal formulations and nutraceutical products.








Similar content being viewed by others
Data availability
All the data utilized for the study were mentioned in the article.
References
Kala CP, Dhyani PP, Sajwan BS (2006) Developing the medicinal plants sector in northern India: challenges and opportunities. J Ethnobiol Ethnomedicine 2(1):1–15. https://doi.org/10.1186/1746-4269-2-32
Selvakumarasamy S, Rengaraju B, Arumugam SA, Kulathooran R (2021) Costus pictus–transition from a medicinal plant to functional food: a review. Future Foods 4:100068. https://doi.org/10.1016/j.fufo.2021.100068
Abirami K, Baskaran V, Singh DR, Gopinath P, Sakthivel K, Roy SD (2014) Phytochemical profile and antifungal activity of Costus sp of Bay Islands. J Andaman Sci Assoc 19(1):45–49
Joseph B, Jacob S, Gupta NK, Binu TK, Benny M, Antony B (2017) Pharmacoepidemiological survey on the use of Costus pictus (insulin plant) in central Kerala. Int J Pharm Pharma Sci 9(4):73–77 https://doi.org/10.22159/ijpps.2017v9i4.16492
Capossio JP, Fabani MP, Reyes-Urrutia A, Torres-Sciancalepore R, Deng Y, Baeyens J, Rodriguez R, Mazza G (2022) Sustainable solar drying of brewer’s spent grains: a comparison with conventional electric convective drying. Processes 10(2):339. https://doi.org/10.3390/pr10020339
Borah A, Hazarika K, Khayer SM (2015) Drying kinetics of whole and sliced turmeric rhizomes (Curcuma longa L.) in a solar conduction dryer. Inf Process Agric 2(2):85–92. https://doi.org/10.1016/j.inpa.2015.06.002
Akpinar EK, Toraman S (2016) Determination of drying kinetics and convective heat transfer coefficients of ginger slices. Heat Mass Transf 52(10):2271–2281. https://doi.org/10.1007/s00231-015-1729-6
Krishnan N, Singh PK, Sakthivelu M, Velusamy P, Gopinath SC, Raman P (2022a) Influence of thermal treatment on extraction and characteristics of phytochemicals from rhizome of Acorus calamus L. Biomass Convers Biorefin:1–16. https://doi.org/10.1007/s13399-022-03415-y
Beigi M, Ahmadi I (2018) Artificial neural networks modeling of kinetic curves of celeriac (Apium graveolens L.) in vacuum drying. Food Sci Technol 39:35–40. https://doi.org/10.1590/fst.35717
Beltramo T, Klocke M, Hitzmann B (2019) Prediction of the biogas production using GA and ACO input features selection method for ANN model. Inf Process Agric 6(3):349–356. https://doi.org/10.1016/j.inpa.2019.01.002
Vinayagam R, Dave N, Varadavenkatesan T, Rajamohan N, Sillanpää M, Nadda AK, Govarthanan M, Selvaraj R (2022) Artificial neural network and statistical modelling of biosorptive removal of hexavalent chromium using macroalgal spent biomass. Chemosphere 296:133965. https://doi.org/10.1016/j.chemosphere.2022.133965
Yasmin T, Asghar A, Ahmad MS, Mehmood MA, Nawaz M (2021) Biorefinery potential of Typha domingensis biomass to produce bioenergy and biochemicals assessed through pyrolysis, thermogravimetry, and TG-FTIR-GCMS-based study. Biomass Convers Biorefin:1–13. https://doi.org/10.1007/s13399-021-01892-1
Krishnan N, Mariappanadar V, Dhanabalan AK, Devadasan V, Gopinath SC, Raman P (2022b) Purification, identification and in silico models of alkaloids from Nardostachys jatamansi—bioactive compounds for neurodegenerative diseases. Biomass Convers Biorefin:1–12. https://doi.org/10.1007/s13399-022-03237-y
Belali M, Seidavi A, Bouyeh M, Gorlov IF, Slozhenkina MI, Mosolov AA, Ramírez LS, Tirado-González DN, Cuevas-Barragán CE, Cipriano-Salazar M (2022) Substantiable bioconversion of the aromatic plant extracts biomass as feed additives in broiler performance: effects and prefeasibility comparison of thyme (Thymus vulgaris). Biomass Convers Biorefin:1–13. https://doi.org/10.1007/s13399-022-02946-8
Aryal S, Baniya MK, Danekhu K, Kunwar P, Gurung R, Koirala N (2019) Total phenolic content, flavonoid content and antioxidant potential of wild vegetables from Western Nepal. Plants 8(4):96. https://doi.org/10.3390/plants8040096
AOAC (1995) Official methods of analysis. In: 16th ed Association of Official Analytical Chemists, Washington, DC, USA.
Asami DK, Hong YJ, Barrett DM, Mitchell AE (2003) Comparison of the total phenolic and ascorbic acid content of freeze-dried and air-dried marionberry, strawberry, and corn grown using conventional, organic, and sustainable agricultural practices. J Agric Food Chem 51(5):1237–1241. https://doi.org/10.1021/jf020635c
Doymaz I (2006) Thin-layer drying behaviour of mint leaves. J Food Eng 74(3):370–375. https://doi.org/10.1016/j.jfoodeng.2005.03.009
Lewis WK (1921) The rate of drying of solid materials. Ind Eng Chem 13(5):427–432. https://doi.org/10.1021/ie50137a021
Onwude DI, Hashim N, Janius RB, Nawi NM, Abdan K (2016) Modeling the thin-layer drying of fruits and vegetables: A review. Compr Rev Food Sci Food Saf 15(3):599–618. https://doi.org/10.1111/1541-4337.12196
Diamante LM, Munro PA (1991) Mathematical modelling of hot air drying of sweet potato slices. Int J Food Sci Technol 26(1):99–109. https://doi.org/10.1111/j.1365-2621.1991.tb01145.x
Midilli A, Kucuk H, Yapar Z (2002) A new model for single-layer drying. Dry Technol 20(7):1503–1513. https://doi.org/10.1081/DRT-120005864
Jafari SM, Ganje M, Dehnad D, Ghanbari V (2016) Mathematical, fuzzy logic and artificial neural network modeling techniques to predict drying kinetics of onion. J Food Process Preserv 40(2):329–339. https://doi.org/10.1111/jfpp.12610
Midilli A, Kucuk H (2003) Mathematical modeling of thin layer drying of pistachio by using solar energy. Energy Convers Manag 44(7):1111–1122. https://doi.org/10.1016/S0196-8904(02)00099-7
Madamba PS, Driscoll RH, Buckle KA (1996) The thin-layer drying characteristics of garlic slices. J Food Eng 29(1):75–97. https://doi.org/10.1016/0260-8774(95)00062-3
Thompson TL, Peart RM, Foster GH (1968) Matllematical simulation of corn drying a new model. Trans ASAE 11(4):582-586. https://doi.org/10.13031/2013.39473
Wang CY, Singh RP (1978) A single drying equation for rough rice. Trans ASAE 11:668–672
Erbay Z, Icier F (2010) A review of thin layer drying of foods: theory, modeling, and experimental results. Crit Rev Food Sci Nutr 50(5):441–464. https://doi.org/10.1080/10408390802437063
Kumar Y, Singh L, Sharanagat VS, Tarafdar A (2021) Artificial neural network (ANNs) and mathematical modelling of hydration of green chickpea. Inf Process Agric 8(1):75–86. https://doi.org/10.1016/j.inpa.2020.04.001
Suarez C, Viollaz P, Chirife J (1980) Diffusional analysis of air drying of grain sorghum. Int J Food Sci Technol 15(5):523–531. https://doi.org/10.1111/j.1365-2621.1980.tb00971.x
Sun J, Hu X, Zhao G, Wu J, Wang Z, Chen F, Liao X (2007) Characteristics of thin-layer infrared drying of apple pomace with and without hot air pre-drying. Food Sci Technol Int 13(2):91–97. https://doi.org/10.1177/1082013207078525
Akgun NA, Doymaz I (2005) Modelling of olive cake thin-layer drying process. J Food Eng 68(4):455–461. https://doi.org/10.1016/j.jfoodeng.2004.06.023
Dincer I, Hussain MM (2002) Development of a new Bi–Di correlation for solids drying. Int J Heat Mass Transf 45(15):3065–3069. https://doi.org/10.1016/S0017-9310(02)00031-5
Demirhan E, Özbek B (2015) Color change kinetics of tea leaves during microwave drying. Int J Food Eng 11(2):255–263. https://doi.org/10.1515/ijfe-2014-0276
Brand-Williams W, Cuvelier ME, Berset CLWT (1995) Use of a free radical method to evaluate antioxidant activity. LWT – Food Sci Technol 28(1):25–30. https://doi.org/10.1016/S0023-6438(95)80008-5
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26(9-10):1231–1237. https://doi.org/10.1016/S0891-5849(98)00315-3
Benzie IF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 239(1):70–76. https://doi.org/10.1006/abio.1996.0292
Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem 269(2):337–341. https://doi.org/10.1006/abio.1999.4019
Kujala TS, Loponen JM, Klika KD, Pihlaja K (2000) Phenolics and betacyanins in red beetroot (Beta vulgaris) root: distribution and effect of cold storage on the content of total phenolics and three individual compounds. J Agric Food Chem 48(11):5338–5342. https://doi.org/10.1021/jf000523q
Miliauskas G, Venskutonis PR, Van Beek TA (2004) Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem 85(2):231–237. https://doi.org/10.1016/j.foodchem.2003.05.007
Tian Y, Zhao Y, Huang J, Zeng H, Zheng B (2016) Effects of different drying methods on the product quality and volatile compounds of whole shiitake mushrooms. Food Chem 197:714–722. https://doi.org/10.1016/j.foodchem.2015.11.029
Saavedra J, Córdova A, Navarro R, Díaz-Calderón P, Fuentealba C, Astudillo-Castro C, Toledo L, Enrione J, Galvez L (2017) Industrial avocado waste: functional compounds preservation by convective drying process. J Food Eng 198:81–90. https://doi.org/10.1016/j.jfoodeng.2016.11.018
Roman MC, Fabani MP, Luna LC, Feresin GE, Mazza G, Rodriguez R (2020) Convective drying of yellow discarded onion (Angaco INTA): modelling of moisture loss kinetics and effect on phenolic compounds. Inf Process Agric 7(2):333–341. https://doi.org/10.1016/j.inpa.2019.07.002
Riveros-Gomez M, Baldán Y, Román MC, Fabani MP, Mazza G, Rodríguez R (2022) Drying and rehydration kinetics of peeled and unpeeled green apple slices (Granny Smith cv). J Environ Sci Health B 57(10):835–847. https://doi.org/10.1080/03601234.2022.2126246
Murthy TPK, Manohar B (2014) Hot air drying characteristics of mango ginger: Prediction of drying kinetics by mathematical modeling and artificial neural network. J Food Sci Technol 51(12):3712–3721. https://doi.org/10.1007/s13197-013-0941-y
Depiver JA, Mallik S (2023) An empirical study on convective drying of ginger rhizomes leveraging environmental stress chambers and linear heat conduction methodology. Agriculture 13(7):1322. https://doi.org/10.3390/agriculture13071322
Fabani MP, Román MC, Rodriguez R, Mazza G (2020) Minimization of the adverse environmental effects of discarded onions by avoiding disposal through dehydration and food-use. J Environ Manage 217:110947. https://doi.org/10.1016/j.jenvman.2020.110947
Nadi F, Tzempelikos D (2018) Vacuum drying of apples (cv. Golden Delicious): drying characteristics, thermodynamic properties, and mass transfer parameters. Heat Mass Transf 54(7):1853–1866. https://doi.org/10.1007/s00231-018-2279-5
Ju HY, Law CL, Fang XM, Xiao HW, Liu YH, Gao ZJ (2016) Drying kinetics and evolution of the sample's core temperature and moisture distribution of yam slices (Dioscorea alata L.) during convective hot-air drying. Dry Technol 34(11):1297–1306. https://doi.org/10.1080/07373937.2015.1105814
Rayaguru K, Routray W (2010) Effect of drying conditions on drying kinetics and quality of aromatic Pandanus amaryllifolius leaves. J Food Sci Technol 47(6):668–673. https://doi.org/10.1007/s13197-010-0114-1
Rodríguez J, Clemente G, Sanjuán N, Bon J (2014) Modelling drying kinetics of thyme (Thymus vulgaris L.): theoretical and empirical models, and neural networks. Food Sci Technol Int 20(1):13–22. https://doi.org/10.1177/1082013212469614
Zalpouri R, Singh M, Kaur P, Kaur A, Gaikwad KK, Singh A (2023) Drying kinetics, physicochemical and thermal analysis of onion puree dried using a refractance window dryer. Processes 11(3):700. https://doi.org/10.3390/pr11030700
Kalsi BS, Singh S, Alam MS (2023) Bhatia S (2023) Microwave drying modelling of Stevia rebaudiana leaves using artificial neural network and its effect on color and biochemical attributes. J Food Qual. https://doi.org/10.1155/2023/2811491
Kaveh M, Sharabiani VR, Chayjan RA, Taghinezhad E, Abbaspour-Gilandeh Y, Golpour I (2018) ANFIS and ANNs model for prediction of moisture diffusivity and specific energy consumption potato, garlic and cantaloupe drying under convective hot air dryer. Inf Process Agric 5(3):372–387. https://doi.org/10.1016/j.inpa.2018.05.003
Markowski M (1997) Air drying of vegetables: evaluation of mass transfer coefficient. J Food Eng 34(1):55–62. https://doi.org/10.1016/S0260-8774(97)00018-6
Zogzas NP, Maroulis ZB, Marinos-Kouris D (1996) Moisture diffusivity data compilation in foodstuffs. Dry Technol 14(10):2225–2253. https://doi.org/10.1080/07373939608917205
Dincer I (1998) Moisture transfer analysis during drying of slab woods. Heat Mass Transf 34(4):317–320. https://doi.org/10.1007/s002310050265
McMinn WAM, Khraisheh MAM, Magee TRA (2003) Modelling the mass transfer during convective, microwave and combined microwave-convective drying of solid slabs and cylinders. Food Res Int 36(9-10):977–983. https://doi.org/10.1016/S0963-9969(03)00118-2
Darvishi H, Farhudi Z, Behroozi-Khazaei N (2017) Mass transfer parameters and modeling of hot air drying kinetics of dill leaves. Chem Prod Process Model 12(2). https://doi.org/10.1515/cppm-2015-0079
Gluesenkamp KR, Boudreaux P, Patel VK, Goodman D, Shen B (2019) An efficient correlation for heat and mass transfer effectiveness in tumble-type clothes dryer drums. Energy 172:1225–1242. https://doi.org/10.1016/j.energy.2019.01.146
Hoque MA, Bala BK, Hossain MA, Uddin MB (2013) Drying kinetics of ginger rhizome (Zingiber officinale). Bangladesh J Agric Res 38(2):301–319 https://doi.org/10.3329/bjar.v38i2.15892
Shiny CT, Saxena A, Gupta SP (2013) Phytochemical investigation of the insulin plant “Costus pictus” D. Don. Int J Pharm Biomed Res 4(2):97–104
Ghafoor K, Al Juhaimi F, Özcan MM, Uslu N, Babiker EE, Ahmed IAM (2020) Total phenolics, total carotenoids, individual phenolics and antioxidant activity of ginger (Zingiber officinale) rhizome as affected by drying methods. LWT 126:109354. https://doi.org/10.1016/j.lwt.2020.109354
Prathapan A, Lukhman M, Arumughan C, Sundaresan A, Raghu KG (2009) Effect of heat treatment on curcuminoid, colour value and total polyphenols of fresh turmeric rhizome. Int J Food Sci Technol 44(7):1438–1444. https://doi.org/10.1111/j.1365-2621.2009.01976.x
Wojdyło A, Figiel A, Lech K, Nowicka P, Oszmiański J (2014) Effect of convective and vacuum–microwave drying on the bioactive compounds, color, and antioxidant capacity of sour cherries. Food Bioproc Tech 7:829–841. https://doi.org/10.1007/s11947-013-1130-8
Marić L, Malešić E, Tušek AJ, Benković M, Valinger D, Jurina T, Kljusurić JG (2020) Effects of drying on physical and chemical properties of root vegetables: Artificial neural network modelling. Food Bioprod Process 119:148–160. https://doi.org/10.1016/j.fbp.2019.11.002
El-Ghorab AH, Nauman M, Anjum FM, Hussain S, Nadeem M (2010) A comparative study on chemical composition and antioxidant activity of ginger (Zingiber officinale) and cumin (Cuminum cyminum). J Agric Food Chem 58(14):8231–8237. https://doi.org/10.1021/jf101202x
Ahmed M, Eun JB (2018) Flavonoids in fruits and vegetables after thermal and nonthermal processing: a review. Crit Rev Food Sci Nutr 58(18):3159–3188. https://doi.org/10.1080/10408398.2017.1353480
Zhu Y, Pu BQ, Xie GY, Tian M, Xu FY, Qin MJ (2014) Dynamic changes of flavonoids contents in the different parts of rhizome of Belamcanda chinensis during the thermal drying process. Molecules 19(7):10440–10454. https://doi.org/10.3390/molecules190710440
Sharma S, Dhalsamant K, Tripathy PP, Manepally RK (2021) Quality analysis and drying characteristics of turmeric (Curcuma longa L.) dried by hot air and direct solar dryers. Lwt 138:110687. https://doi.org/10.1016/j.lwt.2020.110687
Cheaib D, El Darra N, Rajha HN, El-Ghazzawi I, Mouneimne Y, Jammoul A, Maroun RG, Louka N (2018) Study of the selectivity and bioactivity of polyphenols using infrared assisted extraction from apricot pomace compared to conventional methods. Antioxidants 7(12):174. https://doi.org/10.3390/antiox7120174
Author information
Authors and Affiliations
Contributions
SS—Experimental analysis, validation, manuscript (original draft and review). BR—Supervision and review of the manuscript. RK—Conceptualization. AT—Subject expert for statistical tools.
Corresponding author
Ethics declarations
Ethical approval
The research conducted did not involve any studies with animal or human participants, and it was not conducted in any private or protected areas.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Selvakumarasamy, S., Rengaraju, B., Kulathooran, R. et al. Artificial neural network and mathematical modeling on the drying kinetics of Costus pictus rhizomes and its impact on the polyphenol, flavonoid content and antioxidant activity. Biomass Conv. Bioref. (2023). https://doi.org/10.1007/s13399-023-04958-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13399-023-04958-4